|
Refer to for product manuals for complete indications, contraindications, warnings, precautions and potential adverse events. Adverse events may result in fluctuations in blood glucose in patients with diabetes. May include: undesirable change in stimulation (uncomfortable, jolting or shocking) hematoma, epidural hemorrhage, paralysis, seroma, infection, erosion, device malfunction or migration, pain at implant site, loss of pain relief, and other surgical risks. Komoroski C, Roy L: Evaluation of an innovative spinal cord stimulator platform for the treatment of chronic pain. Recharging a rechargeable neurostimulator may result in skin irritation or redness near the implant site. These studies also suggest that the direct activation of dorsal column axons is also unlikely with clinical KHFSCS. Avoid activities that put stress on the implanted neurostimulation system components. Safety and effectiveness has not been established for pediatric use, pregnancy, unborn fetus, or delivery. A preoperative assessment is advised for some patients with diabetes to confirm they are appropriate candidates for surgery. Cameron T: Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20-year literature review. Patients with diabetes may have more frequent and severe complications with surgery. One-year follow-up image revealing dorsal column stimulator paddle lead at C1-C2. An implanted cardiac device (e.g., pacemaker, defibrillator) may damage a neurostimulator, and electrical pulses from the neurostimulator may cause inappropriate response of the cardiac device. The treatment of chronic pain using DRGS has primarily focused on conditions involving severe neuropathic pain including complex regional pain syndrome 5, 6.However, emerging evidence has focused this therapy in the treatment of spine pain, especially difficult to treat low back pain 7, 8. Sources of electromagnetic interference (e.g., defibrillation, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the system, resulting in unexpected changes in stimulation, serious patient injury or death. The mechanism of action of SCS would be based on the antidromic activation of the dorsal column fibers, which activate the inhibitory interneurons within. Its exact role in relation to intrathecal baclofen infusion and ablative procedures remains to be defined.Spinal cord stimulation (SCS) is indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain.ĭiathermy - Energy from diathermy can be transferred through the implanted system and cause tissue damage resulting in severe injury or death. It appears that spinal cord stimulation is an effective and safe alternative in the management of spasms in spinal cord injury victims. 
A statistically significant reduction in the severity of the spasms was observed in the follow-up evaluations, with results that progressively increased in time. 
No patient observed neurological deterioration following the surgical procedure or the neurostimulation treatment. Treatment of Neuropathic Pain and Functional Limitations Associated With Multiple Sclerosis Using an MRI-Compatible Spinal Cord Stimulator: A Case Report With Two Year Follow-Up and Literature Review. Spinal Cord Stimulation (SCS) has been used with. Unfortunately, these medications have many potential side effects and risks. Between 1996-2016, low back and neck pain expenditures amounted to 134.5 billion according to the Journal of the American Medical Association. Opioid-based painkillers are often necessary for chronic pain. It can help a person rely less on stronger pain medications. Combining the frequency and intensity scores into a ‘severity’ score provided a more accurate clinical picture. Disorder of dorsal column stimulator (271969004). The potential uses for spinal cord stimulation include heart-related chest pain, nerve pain, and spine or back pain. A spinal cord stimulator (SCS) or dorsal column stimulator (DCS) is a type of implantable neuromodulation device (sometimes called a 'pain pacemaker') that is used to send electrical signals to select areas of the spinal cord (dorsal columns) for the treatment of certain pain conditions. 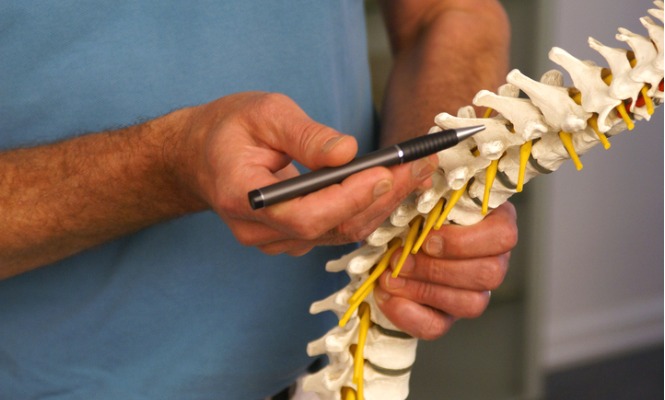
Pre- and postoperative data collection included the frequency and severity of the spasms. Spinal cord stimulation (SCS) is one of the most effective technologies that, unlike other interventional pain treatment methods, achieves long-term results in patients suffering from chronic neuropathic pain. The protocol included assessment by independent examiners preoperatively and at 3, 6, 12 and 24 months after the implant. All the patients were at least 6 months after the injury. Forty-eight spinal cord injury victims were implanted with an epidural spinal cord stimulation system to treat spasms that had not satisfactorily responded to medical therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
